Bacteriological profile of endotracheal tube aspirates in head injury patients admitted in Neurosurgical Intensive Care Unit:a cross-sectional study from a tertiary care hospital of Central Nepal
Joe M.Das ,Sanjana Rajkumari,Shova Dangol,Rashmi SapkotaManish Mishra
1 Department of Neurosurgery,College of Medical Sciences - Teaching Hospital,Bharatpur,Chitwan,Nepal
2 Department of Microbiology,College of Medical Sciences - Teaching Hospital,Bharatpur,Chitwan,Nepal
3 Department of Anesthesiology,College of Medical Sciences - Teaching Hospital,Bharatpur,Chitwan,Nepal
Abstract
Key words:antibiotics;bacteriology;craniocerebral trauma;drug resistance;Intensive Care Units;intratracheal intubation;microbial;Nepal;Neurosurgery;respiratory tract infection;retrospective cross-sectional study
INTRODUCTION
Respiratory tract infection (RTI),especially pneumonia,is a very common disease in Neurosurgical Intensive Care Units (NSICU).Many head injury patients are in an unconscious state and need an endotracheal tube (ETT) (Hubbard et al.,2016).More often they may depend on ventilators for respiratory support (Gianakis et al.,2015).Such patients will be susceptible to infections because of the decreased immunity due to stress,decreased level of consciousness leading to high chance of aspiration,and the presence of artificial tube in the airway (Reden et al.,2006;?ukowicz et al.,2011;Song et al.,2018).Such respiratory infections are a major source of morbidity and mortality in NSICU.Most of the infections are caused by bacteria,especiallyAcinetobacter,Pseudomonas,Escherichia coliandEnterococcus(Kourbeti et al.,2012;Jovanovic et al.,2015).Many gram-negative organisms are usually sensitive tocephalosporins,quinolonesorcarbapenems.But we may have to start the use of an empirical antibiotic when the patient starts to develop an infection which can be changed according to the culture and sensitivity report later.But such reports often take 3-4 days to come and by that time patients' infection may flare-up.So it is important to identify the most common organism causing the infection and its sensitive antibiotic,which can be started initially,but which varies from intensive care unit (ICU) to ICU,hospital to hospital and country to country.Though many studies have been conducted in the western countries regarding the microbiological profile in the endotracheal tube aspirate (ETA) of such patients,literature from developing countries on this subject is scarce and there are only a few such studies conducted in Nepal till date (Ranjit and Bhattarai,2011;Khanal et al.,2013;Khan et al.,2015;Parajuli et al.,2017).No such studies have been conducted in head injury patients in Nepal according to our knowledge and none has attempted to suggest a proper empirical first line antibiotic for hospital-acquired RTIs in intubated patients.
SUBJECTS AND METHODS
Study setting,design and population
We did a hospital-based retrospective cross-sectional study at the NSICU in our hospital.The study period was from January 2018 to June 2018.Our hospital is a 1050-bedded tertiary care medical college providing the highest quality of services to neurosurgery patients.Our NSICU is one of the biggest and busiest care units in Nepal with 12 beds.We may come across all types of head injuries on a daily basis.As a protocol in NSICU,ETA had to be collected and sent for gram staining and culture and sensitivity determination for all head injury patients who are having ETT for ≥ 3 days for assisting respiration,regardless of whether they had a fever nor not.Those reports were collected retrospectively.No predefined sample size was set prior to the inception of the study.Fifty ETA samples collected during the period of 6 months were included in this study.
This study was approved by the Institutional Review Committee of College of Medical Sciences - Teaching Hospital,Bharatpur on November 28,2017 (Additional file 1),as per the guidelines set in standard operating procedure (SOP) of Approval of Health Research by COMSTH-IRC and in accordance with theDeclaration of Helsinki.All patients' guardians signed a statement of informed consent.
Sample collection and laboratory processing
The ETA was collected from the ETT using a 22-inch,14 FG suction catheter connected to a mucus extractor.A length of approximately 24 cm of the catheter was passed through the ETT and secretions were aspirated without instilling saline.The tip of the catheter was made sure to reach just beyond the tip of ETT.The entire procedure was done under aseptic conditions by trained staff nurses.
Samples of ETA thus obtained were processed by standard operating procedure following the manual of American Society for Microbiology in the Department of Microbiology.These samples were inoculated in routine culture media (blood agar,chocolate agar,and MacConkey agar) [Hi-Media Laboratory Ltd.,Mumbai,India].Gram's stain was prepared for morphology-based identification of bacteria.Cultural characteristics and biochemical properties were determined in compliance with the American Society for Microbiology manual.ETA samples were graded following the criteria of Lentino and Lucks.The most purulent or clotted part of ETA was chosen for culture.The agar media were used for quantitative cultures and > 106CFU/mL on quantitative culture was considered pathological (Horan et al.,2008).The plates for bacterial culture were incubated up to 48 hours.Anaerobic cultures were not done in any of these patients.
Antibiotic susceptibility test
Once the bacterial growth was observed,species were identified with the help of Gram's stain and biochemical tests.Antibiotic susceptibility of the bacterial isolates from ETA was performed using a modified Kirby-Bauer disc diffusion method (Midolo et al.,1995) done on Mueller Hinton Agar,supplied by Hi-Media Laboratories and the results were interpreted according to the Clinical &Laboratory Standards Institute guideline.Antibiotic sensitivity tests were performed for 19 antibiotics.The antibiotic discs used were Amikacin (30 μg),Ampicillin (10 μg),Ampicillin/Sulbactam (10 μg /10 μg),Azithromycin (15 μg),Cefotaxime (5 μg),Ceftriaxone (30 μg),Chloramphenicol (30 μg),Ciprofloxacin (5 μg),Ofloxacin (5 μg),Ceftazidime (30 μg),Cefixime (5 μg),Meropenem (10 μg),Cotrimoxazole (25 μg),Gentamicin (120 μg),Cefepime (30 μg),Imipenem (10 μg),Cloxacillin (5 μg),Cefoperazone/Sulbactam (75 μg/30 μg),Piperacillin/Tazobactum (100 μg/10 μg) (Hi-Media Laboratory Ltd.).These antibiotics were chosen due to their commercial availability in Nepal.Some antibiotics were not tested in some organisms due to the known resistance.Multidrug-resistant bacteria were defined as the isolates not susceptible to at least one agent from three different antimicrobial categories of first-line drugs.Some antibiotics were not tested for certain organisms,due to the assumed resistance pattern.We did not include the second-line antibiotics for analysis.
Data analysis
The data obtained were entered in Microsoft Office Excel 2016 and analyzed by Statistical Package for Social Sciences (SPSS) version 25.0 (IBM Corp.,Armonk,NY,USA).We considered the study by Ranjit and Bhattarai (2011) as a baseline regarding the statistical distribution of organisms in order to find out the confidence interval for the sensitivity of antibiotics.Data were reported as the mean ± SD,or cell counts and percentages with 95% confidence intervals.Frequency and percentages were calculated by cell counts and chi-square values andP-values were used to test the significance of attributes between study variables.TheP-value < 0.05 was considered statistically significant.
RESULTS
Fifty samples of ETA were collected from 50 patients,among whom 35 were males and 15 were females.Age of the patients was classified as young (< 45 years),middle-aged (45-65 years) and old (> 65 years).Twenty-three patients had a severe head injury with the Glasgow Coma Scale (GCS) score ≤ 8,16 had a moderate (GCS score > 8 but ≤ 12) and 11 had a mild head injury (GCS score ≥ 13).Among those having a mild head injury,nine patients had to be intubated for the associated injuries or respiratory pathologies and two were intubated for drop in GCS score.There was no statistically significant difference between the distribution of bacteria among the various age groups,genders and different categories of head injury (Table 1).
A total of five different bacteria were isolated from 50 ETA samples,the frequency of which was in the following descending orderAcinetobacter baumanii(18),Pseudomonas aeruginosa(16),Klebsiella pneumonia(8),Escherichia coli(4) andEnterobacterspecies (4) (Figure 1).
Acinetobacter baumaniiwas the most prevalent bacterium causing RTI in NSICU.It was universally resistant to Ampicillin,Ceftriaxone,Cefotaxime,Cefixime and Piperacillin/Tazobactam.Only a very small percentage (5.56%) of the organisms were sensitive to Azithromycin,Ceftazidime,Ciprofloxacin,Ofloxacin,Cloxacillin,and Chloramphenicol.Meropenem (83.3%),Amikacin (44.44%),Cefoperazone/Sulbactam (38.89%) and Ampicillin/Sulbactam (38.89%) were the only antibiotics to which the organism showed good response (Table 2).
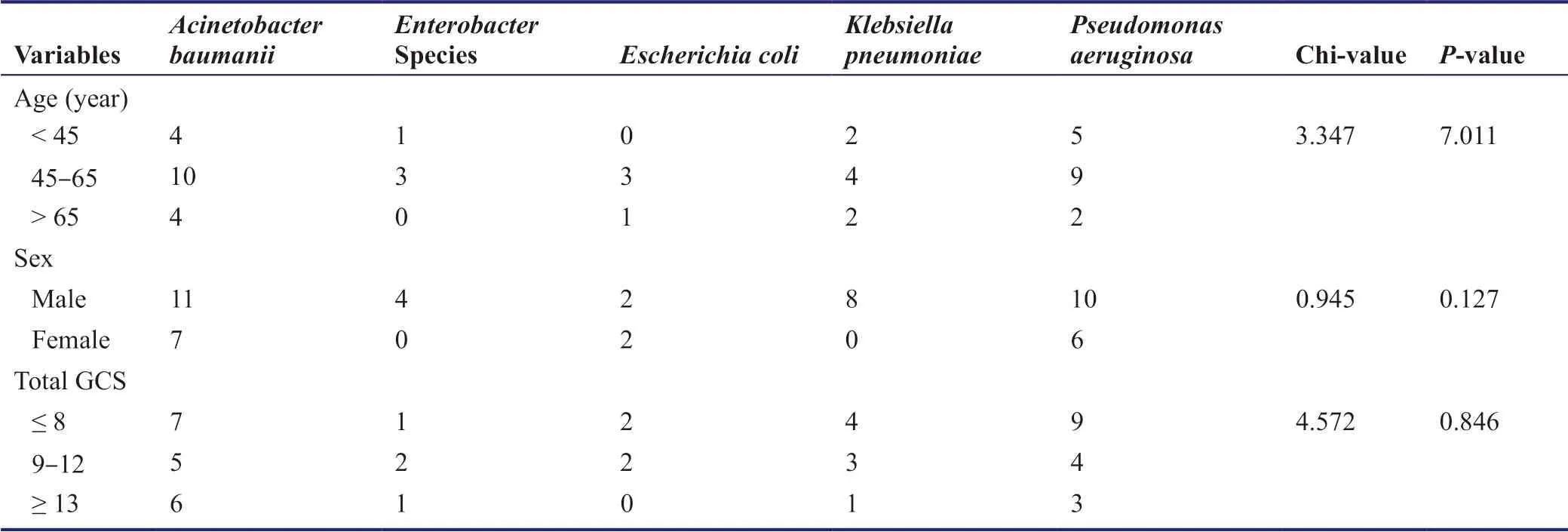
Table 1:The distribution of different bacteria among different age groups,genders and different grades of head injuries
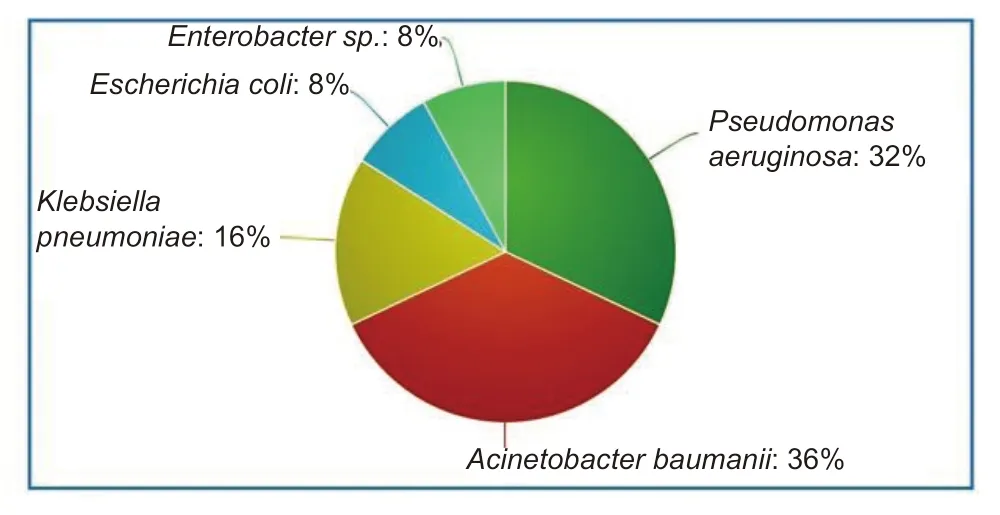
Figure 1:Pie-chart showing the proportion of different species of bacteria in endotracheal aspirates (in percentage).
Pseudomonas aeruginosawas the second most common bacteria in our ICU and it was uniformly resistant to Ampicillin/Sulbactam,Cefixime and Cloxacillin;and maximally sensitive to Meropenem (81.25%),Piperacillin/Tazobactam (75%),Amikacin (68.75%) and Ciprofloxacin (62.5%).It also showed high resistance to Chloramphenicol and Ampicillin with only 6.25% of the organisms responding to the same.
Klebsiella pneumoniawas also found prevalent in ICU,with a high degree of response to Meropenem (50%) and Chloramphenicol (37.5%);and it was 100% resistant to Ampicillin,Ceftriaxone,Ciprofloxacin,Cefotaxime,Cefixime,and Ofloxacin.
Escherichia coliandEnterobacterspecies had equal distributions in our study with both of these being resistant to Ampicillin,Ceftazidime,Ofloxacin,Piperacillin/Tazobactam and Chloramphenicol.WhileEscherichia coliwas 100% sensitive to Amikacin,Cotrimoxazole,Gentamicin,and Meropenem;Enterobacterhad high degrees of drug resistance and was 100% sensitive to Meropenem only.
Most organisms were sensitive to some antibiotics only,many were resistant to all and Meropenem was the only antibiotic which was sensitive to all the organisms (Table 3).
DISCUSSION
RTI is a major cause of morbidity and mortality in ICU.Head injury patients are prone to develop such infections due to the following reasons:
1.Many patients may be in a comatose stage and have ETT.If there is inadequate oral care,pathogens will colonize inside the oral cavity and tongue making the normal secretions highly infectious.These secretions may migrate into the subglottic space and from there micro-leakage occurs into the tracheaviathe small gap between the vocal cords produced by the presence of ETT.
2.Such patients are prone to have a drop in GCS and may require endotracheal intubation at any time.If not intubated on time,oral secretions may be aspirated into the trachea due to loss of protective reflexes.These secretions in the normally sterile airway may act as a reservoir for growth of microbes.
3.Some patients may have to be kept nothing by mouth for a few days,pending the decision for surgery in equivocal cases.It may increase the gastric pH value and volume leading to regurgitation.
4.Many patients may require emergency intubation or reintubation,increasing the chance of infection.
5.Prolonged ventilation,required in many patients,is another proven risk factor.
In multiple studies conducted on ventilator-associated pneumonia and other respiratory infections in several ICUs in Nepal,Acinetobacter sp.was identified as the most common cause (Table 4).Our results support this data withAcinetobacter sp.being responsible for 36% of the infections.The study by Khan et al.(2015) was done in patients with chronic lung diseases.HenceAcinetobacter sp.was not identified in those specimens,which proves that this organism is entirely hospital-acquired.Contrary to other studies in the table,Pseudomonasinfection was found to occur more frequently compared to that due toKlebsiella.There was no case ofStaphylococcusinfection.
Pseudomonasantibiogram taken from a previous study (Parajuli et al.,2017) showed that Ceftazidine (91.6%),Ciprofloxacin (95.8%),Levofloxacin (87.5%),Imipenem (62.5%),Gentamicin (62.5%) were not sensitive and Cotrimoxazole and Tigecycline were sensitive.In our study,Ciprofloxacin and Ceftazidime had only 62.5% and 31.25% sensitivity respectively.Similarly,for Klebsiella,the antibiogram had previously shown that Cefotaxime,Cefepime,and Cotrimoxazole were 100% resistant and the sensitive antibiotics included Penicillin and Tigecycline.Our study showed similar findings regarding drug resistance with 50% sensitivity to Meropenem and 25% sensitivity to Amikacin.
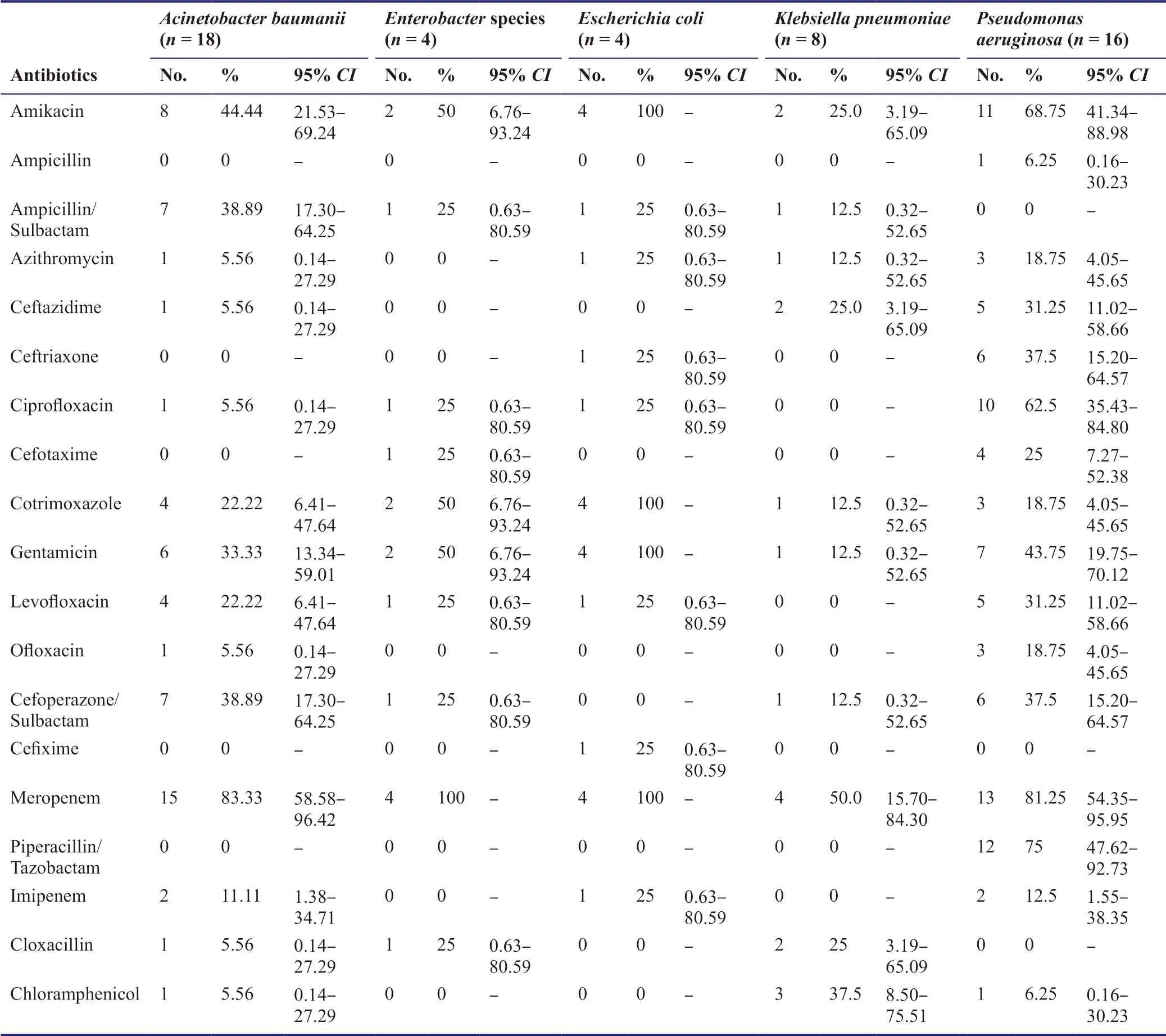
Table 2:The antibiograms of different bacteria isolated from ETAs
A previous review done by Bonell et al.(2019) also found thatAcinetobacter baumanniiwas the primary organism associated with ventilator-associated pneumonia and that it was the commonest organism in low-income countries like Nepal.The rates of carbapenem resistance inAcinetobacter baumanniiwas reported to be approximately 75% as per this review.But our study revealed that even though the organism was resistant to Imipenem,it was highly sensitive to Meropenem,both being Carbapenems.
Multidrug resistance (MDR) is defined as insensitivity or resistance of a microorganism to the administered antimicrobial medicines (which are structurally unrelated and have different molecular targets) despite earlier sensitivity to it (Singh,2013).MDR organisms including those organisms identified in our study are considered as a clinical threat to human and animals (Kallel et al.,2005;Wood et al.,2010).Centers for Disease Control and Prevention (2013) estimated that antibiotic resistance is responsible for more than 2,000,000 infections and 23,000 deaths each year in the United States.According to it,multidrug-resistantAcinetobacterandPseudomonasare considered as serious threats of antibiotic resistance causing around 500 and 440 deaths/year respectively.An estimated 51,000 healthcare-associatedPseudomonas aeruginosainfections occur in the United States every year.More than 13% of these are MDR,with roughly 400 deaths/year caused due to these infections.

Table 3:Antibiotics and their corresponding sensitive organisms
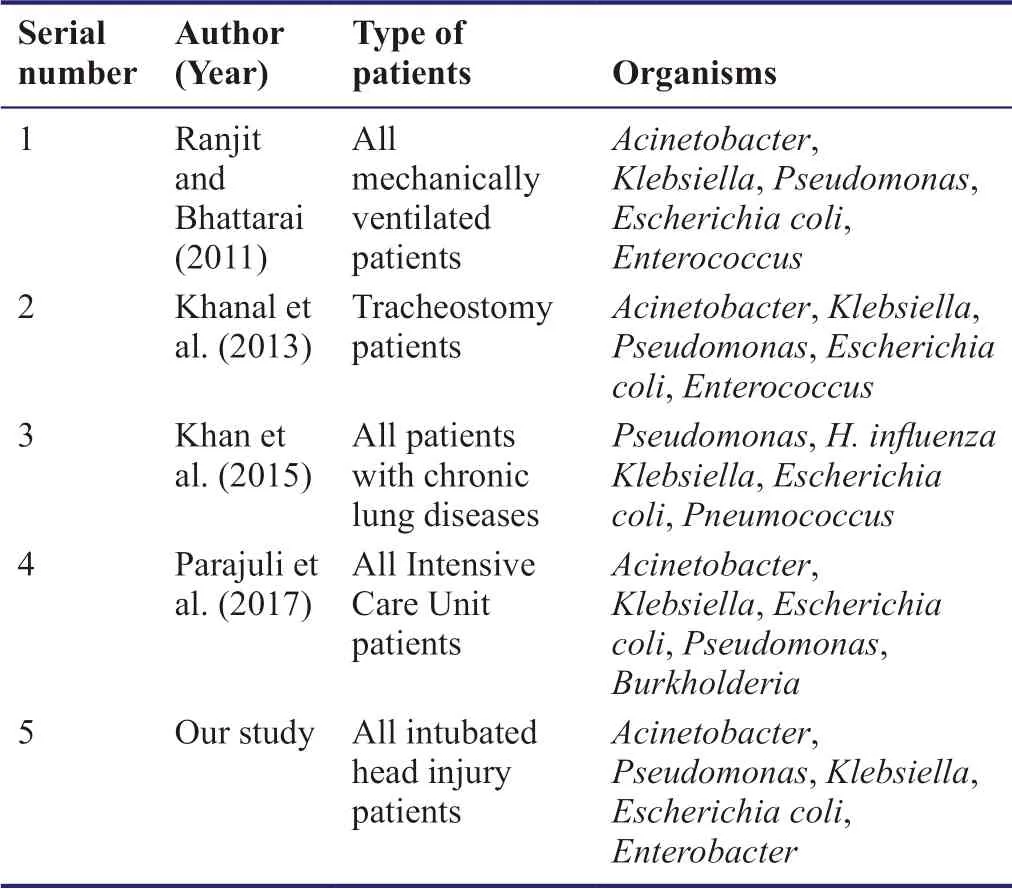
Table 4:The frequency of lower respiratory tract infections caused by different species of bacteria in hospitals of Nepal
Mechanisms of MDR(Tanwar et al.,2014):
1.Increased drug efflux from the cells;
2.Altered membrane permeability of the bacterial cells;
3.Intracellular degradation and inactivation of drugs by enzymes;
4.Overexpression of drug target thereby inducing DNA repair;
5.Altered drug target by mutation.
Limitations of the study:
1.We have not included the outcome of the patient.
2.The data do not include which antibiotic was started for the patients and what the clinical response was.
3.Coexisting respiratory disease or chest injury,which can act as a confounding factor,has not been considered.
4.Data regarding mechanical ventilation was not taken into consideration.
5.Second-line antibiotic sensitivity was not included in the data.
Conclusion
This study identified the main five causative organisms of lower respiratory tract infections in head injury patients in our NSICU,of whichAcinetobacter baumaniiwas the most common.Most of the organisms are multi-drug resistant.Meropenem is found to be the antibiotic sensitive to most of the organisms,making it the drug of choice to be started empirically.Amikacin is to be the second choice since it is found to be sensitive toEscherichia coli,PseudomonasandAcinetobacter.
Additional file
Additional file 1:Hospital Ethics Approval.
Acknowledgments
We would like to acknowledge the services of Mr.Rakesh Basnyet,Health Assistant,in collecting the data.
Author contributions
All authors contributed to the study design,data analysis,and paper writing,and approved the final paper.
Conflicts of interest
The authors have no conflicts of interest to declare.
Financial support
None.
Institutional review board statement
This study was approved by the Institutional Review Committee of College of Medical Sciences - Teaching Hospital,Bharatpur on November 28,2017 as per the guidelines set in standard operating procedure (SOP) of Approval of Health Research by COMSTH-IRC and in accordance with theDeclaration of Helsinki.All patients signed a statement of informed consent.
Declaration of participant consent
The authors certify that they have obtained all appropriate patient consent forms.In the form the patients' guardians have given their consent for the patients' images and other clinical information to be reported in the journal.The patients' guardians understand that the patients' names and initials will not be published and due efforts will be made to conceal their identity.
Reporting statement
This study followed the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement.
Biostatistics statement
The statistical methods of this study were reviewed by the statisticians of College of Medical Sciences - Teaching Hospital,Bharatpur,Nepal.
Copyright license agreement
The Copyright License Agreement has been signed by all authors before publication.
Data sharing statement
Individual participant data that underlie the results reported in this article,after deidentification (text,and tables),will be available upon request.Data will be available immediately following publication,no end date for anyone who wishes to access the data.In order to gain access,data requestors will need to sign a data access agreement.Proposals should be directed to drjoemdas@gmail.com.
Plagiarism check
Checked twice by iThenticate.
Peer review
Externally peer reviewed.
Open access statement
This is an open access journal,and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License,which allows others to remix,tweak,and build upon the work non-commercially,as long as appropriate credit is given and the new creations are licensed under the identical terms.
 Asia Pacific Journal of Clinical Trials:Nervous System Diseases2019年3期
Asia Pacific Journal of Clinical Trials:Nervous System Diseases2019年3期
- Asia Pacific Journal of Clinical Trials:Nervous System Diseases的其它文章
- Effectiveness of inpatient alcohol detoxification and psychotherapeutic support program:a prospective self-controlled study at a tertiary hospital in Nepal
- Mindfulness-based cognitive therapy for university students with depression,anxiety,and stress symptoms:a randomized controlled trial
- Treatment with a halved dose of antipsychotics in patients with schizophrenia who relapse while receiving high-dose antipsychotic therapy
